In 1999-2001, I developed a dissociated Zebrafish spinal neuron culture system inspired by dissociated Xenopus spinal neuron cultures (Zebrafish spinal neuron culture paper (2001) [PDF]). In vitro use of Zebrafish has since become increasingly applied in research amongst the various model systems available. The concept of ‘model system’ is fairly new in biological science. It arose with the development of molecular biology and the adoption of a reductionist approach. It is now common to observe groups of scientists working on the same scientific questions but in different ‘model systems’. For various reasons some organisms became popular models, and this trend was further promoted by the structuring of the scientific community and funding agencies, as well as the academic ‘laboratory-lineage’ hiring policies [1]. For basic developmental and neurobiology studies, two organism that became widely used as models, and still are today, were Drosophila melanogaster and Caenorhabditis elegans. Although there are compelling reasons to work with these model organisms, they are border-liners when it comes to neurobiology because of their rudimentarily developed nervous system, and their lack of an actual brain.
Because of the potential of Zebrafish for developmental biology, and for reasons amply stated in this issue, George Streisinger in the mid-1970s started work on this vertebrate [2]. Now at the dawn of the 21st century, it is clear that Zebrafish is here to stay as model organism, and that its contribution to the fields of developmental and neurobiology will become comparable to that of Drosophila in the 20th century. The Zebrafish model evolved from the traditions and approaches of the developmental biologists. Thus, most studies carried out so far have been performed in vivo, and with minimal reliance on in vitro approaches or manipulations other than genetics. The publication in 1996 of an issue of the journal Development [3] marks a milestone in the development of Zebrafish as an experimental model. That issue describes the identification of thousands of mutants as well as their more or less detailed assignment to specific parts of the Zebrafish. However, it is clear that Zebrafish has potential beyond that, and with this in mind, I composed this first Zebrafish issue of Methods in Cell Science.
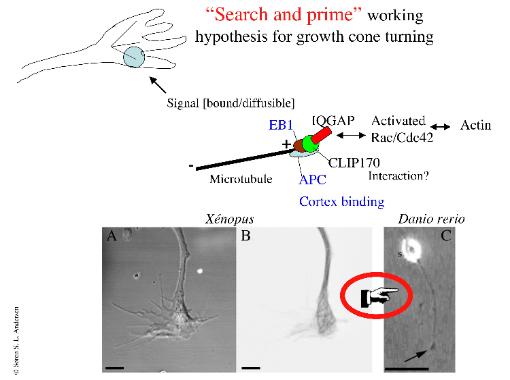
In this issue you can read how the use of various green-fluorescent protein variants, in combination with in vitro and in vivo genetic methods, provides a powerful approach for questions related to neural fate analysis, axon guidance and synapse formation. Related to these areas, a few years ago I was filming GFP-microtubules in dissociated Xenopus laevis spinal neuron cultures (Figure 1). However, to adequately attack such cell biological areas and questions, I developed a Zebrafish culture system [4] with the aim of moving from the Xenopus to the Zebrafish system, where one more readily can use a combinedin vitro and in vivo approach (illustrated by the pointing hand in Figure 1). Further in this issue, a couple of papers illustrate how to obtain electrophysiological recordings from in vitro or in vivo preparations. The study of regeneration following nerve injury has significantly progressed in recent years. Unlike mammals, Zebrafish are capable of regenerating CNS neurons following injury, and a series of papers address this issue through application of various labeling and immunocytochemistry...